all locations
- alabama
- arkansas
- california
- colorado
- connecticut
- district of columbia
- delaware
- florida
- georgia
- iowa
- illinois
- indiana
- kansas
- kentucky
- massachussetts
- maryland
- michigan
- minnesota
- missouri
- mississippi
- north carolina
- nebraska
- new hampshire
- new jersey
- nevada
- new york
- ohio
- oklahoma
- oregon
- pennsylvania
- rhode island
- south carolina
- tennessee
- texas
- virginia
- washington
- wisconsin
branch per city
-
A-D
- altamonte springs, FL
- andover, MA
- aston, PA
- athens, GA
- atlanta, GA
- austin, TX
- bakersfield, CA
- baltimore, MD
- bartlett, TN
- beaverton, OR
- bedford, NH
- bellevue, WA
- bel air, MD
- birmingham, AL
- bloomington, MN
- bolingbrook, IL
- boston, MA
- bowling green, KY
- brunswick, GA
- calhoun, GA
- canton, MA
- carle place, NY
- carrollton, GA
- chambersburg, PA
- charlotte, NC (south college street)
- charlotte, NC (ayrsly town blvd)
- charlotte, NC (north tryon street)
- chattanooga, TN
- cincinnati, OH
- city of industry, CA
- clarksville, TN
- clearwater, FL
- cleveland, TN
- columbus, OH
- conyers, GA
- cookeville, TN
- cranbury, NJ
- dalton, GA
- denver, CO
- duluth, GA
- durham, NC
-
E-H
-
I-M
- independence, OH
- irvine, CA
- irving, TX
- islandia, NY
- jacksonville, FL (saint johns avenue)
- jacksonville, FL (touchton road east)
- johnson city, TN
- kent, WA
- knoxville, TN
- lakeland, FL
- lanham, MD
- las vegas, NV
- lathrop, CA
- lawrenceville, GA
- lenexa, KS
- lexington, KY
- liberty, MO
- lithia springs, GA
- louisville, KY
- macon, GA
- madisonville, TN
- maple grove, MN
- marlborough, MA
- mclean, VA
- mechanicsburg, PA
- melville, NY
- memphis, TN (poplar avenue)
- memphis, TN (winchester road)
- miami lakes, FL
- milledgeville, GA
-
N-R
- nashville, TN
- new york, NY
- north charleston, SC
- north haven, CT
- oakland, CA
- oklahoma city, OK
- omaha, NE
- ontario, CA
- orlando, FL
- ormond beach, FL
- parsippany, NJ
- peachtree city, GA
- phoenix, AZ
- plainville, CT
- plano, TX
- pooler, GA
- porterville, CA
- pulaski, TN
- raleigh, NC
- roanoke, VA
- rocky mount, NC
- rome, GA
- roseville, MN
-
S-Z
- saint louis, MO
- san antonio, TX
- san diego, CA
- san francisco, CA
- san jose, CA
- san mateo, CA
- seven fields, PA
- shelbyville, TN
- southampton, PA
- spartanburg, SC
- stockbridge, GA
- sugar land, TX
- tallahassee, FL
- tampa, FL
- tempe, AZ
- troy, MI
- tucson, AZ
- tullahoma, TN
- warren, OH
- washington, DC
- washington court house, OH
- wauwatosa, WI
- wayne, PA
- west memphis, AR
- west palm beach, FL
- wichita, KS
- williamsville, NY
- winston-salem, NC
- woburn, MA
workforce solutions for employers
-
staffing solutions that fits your needs.
No matter your talent needs, be it temporary, permanent, or contract positions - Randstad is here to help you find the perfect fit. Our nationwide reach ensures that we can locate the ideal staff for your specific requirements, across all industries and qualification levels.
-
operational talent solutions.
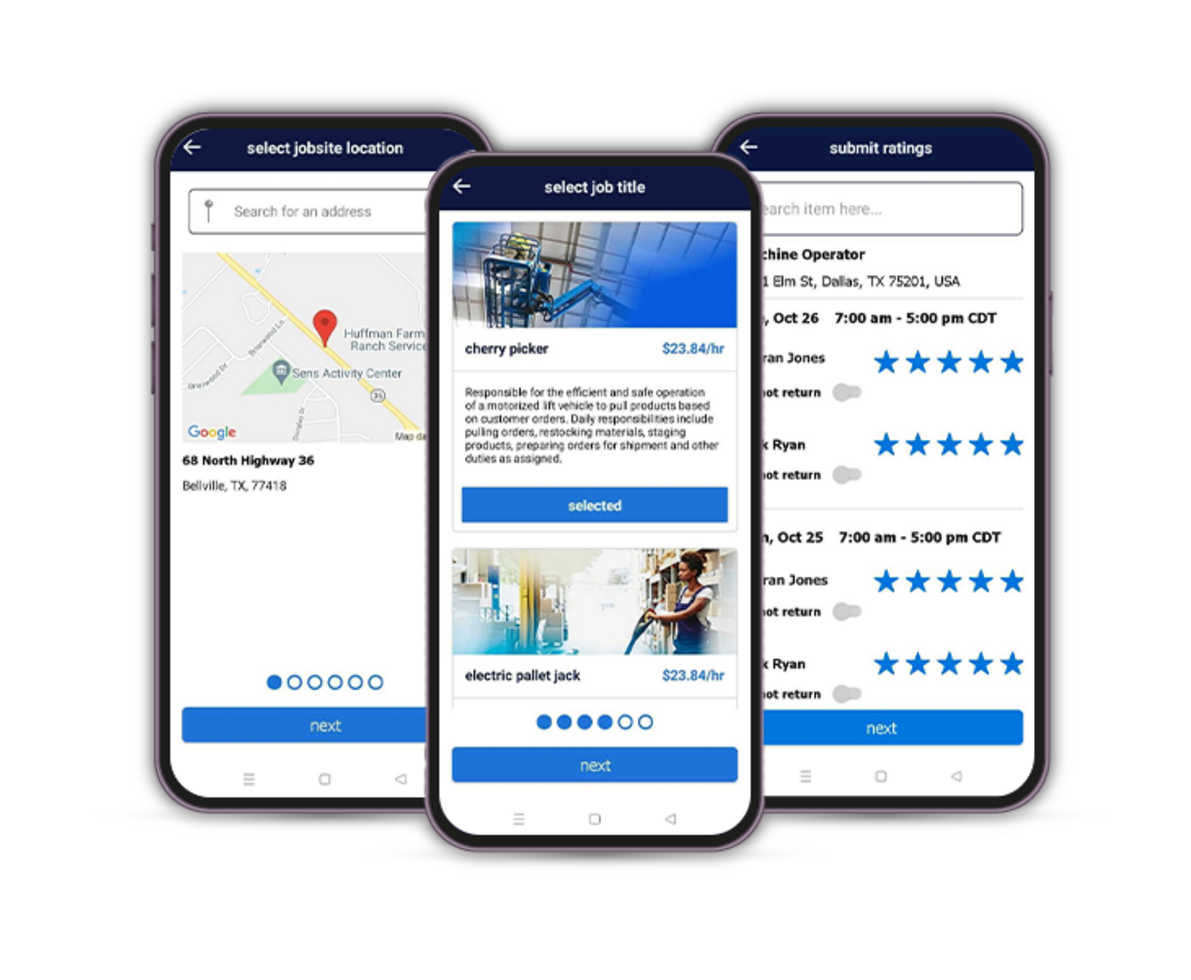
Build a high-performing workforce with qualified, job-ready talent. With access to one of the largest pools of pre-vetted candidates, deep industry expertise, and proven validation processes led by our specialized consultants, we help you simplify hiring and scale with speed and confidence. Need talent fast? Our digital talent marketplace—the Randstad App—connects you to available workers in real time, 24/7.
-
professional talent solutions.
Build your team with top operational talent. With access to the largest pool of pre-qualified candidates, deep industry expertise, and proven talent validation processes in our specialized talent centers, we’ll help you simplify recruiting and scale your workforce quickly and cost-effectively.
frequently asked questions
contact randstad.